Author: Admin
May 2023: New Test – Clozapine (CLOZA)
Clinical Updates
New Test: Clozapine (CLOZA)
Therapeutic drug monitoring for clozapine will be offered as an in-house test starting Tuesday, May 23, 2023.
The current send-out test, Clozapine and Metabolites, Serum or Plasma, Quantitative (CLOZSP), currently sent out to ARUP Laboratories, will be discontinued on June 20, 2023.
Clozapine (CLOZA)
New Test – CLOZA
Specimen Type
Red Serum Tube (No Additive)
Methodology
Turbidimetric immunoassay
Result Components
Clozapine
Days Performed
Monday – Saturday
Turnaround Time
1-4 days
Sendout Test – CLOZSP
Specimen Type
Red Serum Tube (No Additive) or Lavender K2EDTA Tube
Methodology
Liquid chromatography-tandem mass spectrometry
Result Components
Clozapine
Norclozapine
Clozapine-N-Oxide
Days Performed
Sunday – Saturday
Turnaround Time
2-4 days
May 2023: COVID-19 Testing – CPT Code Update
Immediate Test Change
COVID-19 Testing – CPT Code Update
In accordance with recent guidance provided by the Department of Health and Human Services on May 9, 2023, the CPT code associated with COVID-19 testing will change.
Beginning May 11, 2023, the CPT code is changing as a result of the end of the COVID-19 Public Health Emergency.
The new CPT code will display in the Test Directory starting May 11, 2023.
COVID-19 Testing
New CPT Code
87635 (previous: U0003)
Additional Information
For more information, please refer to HHS’s Fact Sheet: End of the COVID-19 Public Health Emergency.
May 2023: Changes to Legionella Diagnostic Testing (LEGPCR)
Clinical Updates
Changes to Legionella Diagnostic Testing (LEGPCR)
Improvements to Cleveland Clinic Laboratories’ Legionella PCR assay will result in the discontinuation of most Legionella cultures.
Beginning May 2, 2023, Legionella pneumophila PCR (LEGPCR) will include additional DNA targets for Legionella, including species other than Legionella pneumophila, and a target specific for Legionella pneumophila serogroup 1.
This assay design is modeled after the U.S. Centers for Disease Control & Prevention’s assay.1
Legionella pneumophila PCR (LEGPCR)
Specimen Requirements
Lower respiratory samples, including bronchoalveolar lavage (BAL) and sputa, will continue to be the acceptable specimen types for PCR.
Results
Possible interpretations of test results include:
- Legionella pneumophila Serogroup 1 Detected
- Legionella pneumophila (not Serogroup 1) Detected
- Legionella species (not pneumophila) Detected
Additional Updates
Beginning May 23, 2023, routine culture for Legionella spp. (LEGCUL) will be discontinued, but culture will reflexively be performed on any sample with detectable Legionella DNA by PCR.
Note: There is no change to Legionella urine antigen (LEGUAG) testing.
Reference
1 Benitez AJ, Winchell JM. Clinical application of a multiplex real-time PCR assay for simultaneous detection of Legionella species, Legionella pneumophila, and Legionella pneumophila serogroup 1. J Clin Microbiol. 2013 Jan;51(1):348-51. doi: 10.1128/JCM.02510-12. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3536254/pdf/zjm348.pdf
April 2023: Updates to H. pylori Urea Breath Test
Immediate Test Change
Updates to H. pylori Urea Breath Test (HPYLBR)
Updated Test
Helicobacter pylori Breath Test (HPYLBR)
Effective May 2, 2023, the Immunopathology Laboratory section of Cleveland Clinic Laboratories will provide a new H. pylori urea breath test.
The qualitative test, based on infrared spectroscopy using 13C, is an aid in diagnosing active infection with Helicobacter pylori and post-treatment monitoring in adult and pediatric patients ages 3-17 years old.
Changes to Specimen Collection
Collection Kits
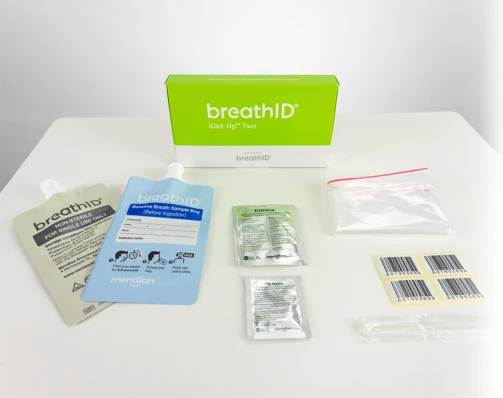
The BreathID® Hp® Two test requires specimen collection with the BreathID® IDkit Hp® Two kit.
One box contains five kits. Each kit contains:
- (1) Blue baseline breath collection bag
- (1) Grey post-ingestion breath collection bag
- 75mg 13C-urea tablet
- 4.3g package of Citrica (citric acid)
- Straw
- Drinking cup
- User guide
- (4) Labels
- Sample transport bag
- Package insert
Test Overview
Test Name
Helicobacter pylori Breath Test
Test Code
CPT Code
83013
Methodology
13C Infrared Spectroscopy
Specimen Type
1 baseline (blue) and 1 post-dose (gray) breath sample bag collected with a BreathID® IDkit Hp® One kit
Stability
Ambient
14 days
Refrigerated
Unacceptable
Frozen
Unacceptable
Reference Range
Negative
Clinical Information
The Urea Breath Test is used as an aid in the diagnosis of current infection with Helicobacter pylori.
Limitations
Despite very high specificity, false positive results may occur due to other gastric organisms, such as H. heilmanni, as well as in patients with hypo- or achlorhydria.
False-negative results may occur in patients who have received antibiotics, proton pump inhibitors, or bismuth preparations.
Clinical correlation is required.
February 2023: Send-out Test Discontinuation – T3 Uptake (T3U)
December 2022: Changes to Acceptable Tube Type for Azole Anti-Fungal Therapeutic Drug Monitoring
Special Communication
December 2022: Changes to Acceptable Specimen Containers for Azole Anti-Fungal Therapeutic Drug Monitoring
Effective December 20, 2022
Plasma collected in Green Lithium Heparin Tubes will no longer be accepted for azole anti-fungal therapeutic drug monitoring tests.
Please collect samples for Azole Anti-Fungal Therapeutic Drug Monitoring tests in Red (serum) No Additive Tubes, centrifuge, and transfer the serum to an aliquot tube.
Tests Affected:
- Fluconazole (FLUC)
- Isavuconazole (ISACON)
- Itraconazole (ITRAC)
- Posaconazole (POSACN)
- Voriconazole (VORCON)
Updated Specimen Requirements
Specimen Type:
Serum
Volume:
0.5 mL
Collection Container:
Red (Serum) No Additive Tube
Transport Temperature:
Refrigerated
Pathology Insights: Tumors of the Lung with Sanjay Mukhopadhyay, MD
Pathology Insights Video Series
Tumors of the Lung
Presented by Sanjay Mukhopadhyay, MD
In this video, an expert pulmonary pathologist and director of Cleveland Clinic’s Pulmonary Pathology service briefly covers all lung tumor types – including all the subtypes of lung cancer – listed in the new (2021) World Health Organization classification. Dr. Mukhopadhyay then describes the primary pathologic features of these tumors with high-quality pathology images provided in most cases.
This video should be helpful for patients, surgeons, pulmonologists, oncologists, pathologists, residents, and medical students.
As part of our educational mission for our clients and communities, Cleveland Clinic Laboratories presents the Pathology Insights video series.
These short videos break down information about interesting pathology cases to better inform doctors, laboratory staff, patients, or anyone interested in the field of pathology. Each episode features important cases, methods, and practices that are personally presented by our staff pathologists.
August 2022: Changes to C Telopeptide, Beta Cross Linked (CTELO)
Special Communication
August 2022: Changes to C Telopeptide, Beta Cross Linked (CTELO)
Effective September 6, 2022
C Telopeptide, Beta Cross Linked (CTELO)
Changes to Specimen Requirements
Specimen Type:
Serum
Volume:
1 mL
Minimum Volume:
0.5 mL
Collection Container:
Gold Serum Separation Tube (SST)
Transport Temperature:
Frozen, critical
Collection Information:
Allow the tube to sit for 15-20 minutes at room temperature to form a clot. Centrifuge and separate serum from cells ASAP or within 2 hours of collection. Transfer serum to a standard aliquot tube.
Alternative Specimen
Alternative Specimen Type:
Plasma
Volume:
1 mL
Minimum Volume:
0.5 mL
Collection Container:
Lavender K2EDTA Tube
Transport Temperature:
Frozen, critical
Collection Information:
Centrifuge and separate plasma from cells ASAP or within 2 hours of collection. Transfer plasma to a standard aliquot tube.
Changes to Reference Ranges
Male
6 Months to 6 Years:
500 – 1700 pg/mL
7 to 9 Years:
522 – 1682 pg/mL
10 to 12 Years:
553 – 2071 pg/mL
13 to 15 Years:
485 – 2468 pg/mL
16 to 17 Years:
276 – 1546 pg/mL
18 to 29 Years:
238 – 1019 pg/mL
30 to 39 Years:
225 – 936 pg/mL
40 to 49 Years:
182 – 801 pg/mL
50 to 59 Years:
161 – 737 pg/mL
60 to 69 Years:
132 – 752 pg/mL
70 to 99 Years:
118 – 776 pg/mL
September 2022: Best Practice for Detecting Hyperhomocysteinemia – Homocysteine Testing—Not MTHFR Genotyping
Best Practice for Detecting Hyperhomocysteinemia: Homocysteine Testing—Not MTHFR Genotyping
There is no conclusive evidence supporting the clinical value of MTHFR polymorphism genotyping.
Practice guidelines from multiple professional societies agree that MTHFR polymorphism genotyping should not be ordered as part of clinical evaluation.
If there is a clinical concern regarding hyperhomocysteinemia, Cleveland Clinic Laboratories recommends Homocysteine testing (HOMCYS) in place of MTHFR genotyping. Homocysteine testing is less expensive, generates a result more quickly, and provides additional actionable information for patient management.
Upcoming Changes
Homocysteine (HOMCYS) Test Details
Test Name
Homocysteine
Test Code
CPT Code
83090
Performing Laboratory
Cleveland Clinic Laboratories
FDA Compliance
In Vitro Diagnostic (IVD)
Methodology
Enzymatic
Days Performed
Sun – Sat
Turnaround Time
8 hours
Specimen Requirements
Type:
Plasma
Volume:
1 mL
Specimen Container:
Light Green Lithium Heparin Plasma Separator Tube (PST)
Transport Temperature:
Refrigerated
Alternative Specimen Requirements
Type:
Serum
Volume:
1 mL
Specimen Container:
Gold Serum Separation Tube (SST)
Transport Temperature:
Refrigerated
Special Instructions
• Centrifuge and separate plasma/serum from cells less than one hour after collection.
• If collected in a non-gel separator tube, centrifuge and transfer plasma/serum to a CCL tube and refrigerate.
Stability
Ambient:
4 days
Refrigerated:
4 weeks
Frozen:
10 months
Reference Interval
18-99 Years – Normal:
<15.1 umol/L
Lack of Evidence for MTHFR Polymorphism Genotyping
Recommendation: Test Plasma Homocysteine Levels
There is no conclusive evidence supporting the clinical value of MTHFR polymorphism genotyping.
If there is a clinical concern regarding hyperhomocysteinemia, Cleveland Clinic Laboratories recommends Homocysteine (HOMCYS) testing.
• Multiple practice guidelines agree that MTHFR polymorphism genotyping should not be ordered as part of a clinical evaluation.
• A cheaper, faster, and more accurate way to test for hyperhomocysteinemia is to measure plasma homocysteine levels.
• If plasma homocysteine levels are high, patients can supplement with vitamins such as B6, B12, folate, and folic acid.
• If plasma homocysteine levels are normal, no treatment is indicated—even if there is an MTHFR variant.
Professional Societies with MTHFR Polymorphism Testing Guidelines:
After reviewing these guidelines,
Cleveland Clinic’s RT-PLMI Section of Molecular Pathology, Genomic Medicine Institute, and Laboratory Stewardship Committee agreed to discontinue MTHFR polymorphism genotyping.
Background
The MTHFR gene (OMIM: 607093) on 1p36.22 encodes the 5,10-methylenetetrahydrofolate reductase enzyme, which converts 5,10-methylenetetrahydrofolate to 5-methyltetrahydrofolate, the primary circulatory form of folate. This enzyme is also involved in the metabolism of the amino acid, homocysteine. A deficiency of the enzyme can lead to HHC.
Polymorphisms are common variants within a gene that do not necessarily affect its function, unlike pathogenic or disease-causing variants. Two commonly tested polymorphic variants in MTHFR are:
c.665C>T* (p.Ala222Val)
*Historically referred to as C677T, the ‘thermolabile’ variant
c.1286A>C (p.Glu429Ala)
These variants are so common that approximately 25% of individuals with Hispanic ancestry and 15% of North Americans with European ancestry have two copies of c.665C>T.
The presence of two copies of c.665C>T (homozygosity) may result in decreased MTHFR enzyme activity and mild HHC. Neither of these MTHFR polymorphisms causes severe MTHFR deficiency (<20% enzyme activity).
References
1. Lack of Evidence for MTHFR Polymorphism Testing. ACMG Practice Guideline. Genet Med. 2013;15(2):153-6.
2. Inherited Thrombophilias in Pregnancy. ACOG Practice Bulletin. No. 197. American College of Obstetricians and Gynecologists. Obstet Gynecol. 2018;132:e18–34.
3. Levin BL, Varga E. MTHFR: Addressing Genetic Counseling Dilemmas Using Evidence-Based Literature. J Genet Counsel. 2016;25:901-11.
4. Choosing Wisely® Initiative https://www.choosingwisely.org/
5. Eng, C. A Genetic Test You Don’t Need: Testing MTHFR is usually unnecessary. Cleveland Clinic Health Essentials. https://health.clevelandclinic.org/a-genetic-test-you-dont-need/. Accessed November 19, 2020.
Updating Best Practices
November 15, 2022: MTHFR genotyping will no longer be available as a send-out test, even if ordered as a miscellaneous test.
March 16, 2021: MTHFR genotyping will no longer be performed in-house at Cleveland Clinic.