Immediate Test Change
Product Discontinuation: BD CultureSwab™ Liquid Amies Double Swab
Replacements: eSwab™ or Cepheid Collection Device
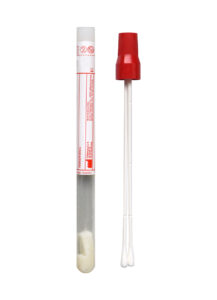
The eSwab™ (Copan Diagnostics) collection device—utilized by Cleveland Clinic for 8+ years—will now be the primary swab for bacteriology.
No change to the swabbing collection process is needed when using an eSwab™.
Although typically tissue or fluid is a superior quality sample for bacteriology, collecting a swab is often more feasible. The eSwab™ uses a patented flocked swab, which enables the elution of the primary sample into 1 mL of liquid contained within the collection tube. The bacteriology laboratory then uses this liquid for testing.
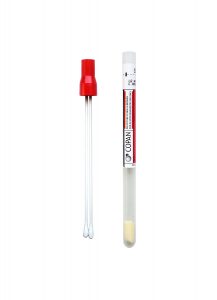
Choose the appropriate collection device:
eSwab™
Use for:
Group A Strep PCR GASPCR
Fungal Screen for Candida FUNGSC
Bacteriology cultures:
• Abscess & Wound Culture WCUL
• Anaerobe Culture ANACUL
• Cystic Fibrosis Respiratory Culture CFRCUL
• Ear Culture & Stain EARCSM
• Eye Culture & Stain EYECSM
• MRSA culture screen MRSASC
• MRSA/S. aureus culture screen SANSAL
• Throat Culture THRCUL
• VRE Culture VRESC