Test Name
Thyroid Stimulating Immunoglobulin (TSIGIM)
CPT Code
84445
Methodology
Chemiluminescent Immunoassay (CLIA)
Turnaround Time
1 – 4 days
Specimen Requirements
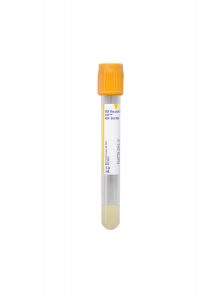
Type:
Serum
Volume:
0.5 mL
Minimum Volume:
0.35 mL
Specimen Container:
Gold BD Hemogard™ Serum Separation Tubes (SST)™
Transport Temperature:
Refrigerated
Alternative Specimen
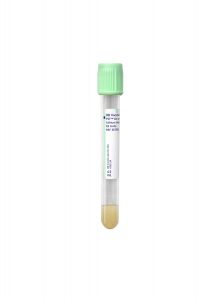
Type:
Plasma
Volume:
0.5 mL
Minimum Volume:
0.35 mL
Specimen Container:
Light Green Lithium Heparin Plasma Separator Tube (PST)
Transport Temperature:
Refrigerated
Stability
Ambient:
24 hours
Refrigerated:
7 days
Frozen:
1 year (≤ -20°C)
Reference Range
<0.55 IU/L
Linear Range
0.10–40.00 IU/L
Clinical Information
The Thyroid Stimulating Immunoglobulin test is used as an aid in diagnosis of autoimmune hyperthyroidism, especially in patients with Grave’s orbitopathy and dermopathy. Low positive TSH receptor stimulating antibody levels may occasionally be found in patients with autoimmune hypothyroidism. Clinical correlation is required.
Additional Information
Background Information
Grave’s disease (GD) is the most common cause of hyperthyroidism. The clinical manifestations are protean and, to some extent, shared with those of Hashimoto thyroiditis (HT), which is a type of autoimmune hypothyroidism. The shared manifestations may include: muscle weakness, menstrual disturbances, hair and skin changes, decreased concentration, fatigue, goiter, and depression.
The autoimmune nature of Grave’s disease was established when the so-called long-activating thyroid stimulator (LATS) in sera from patients with Grave’s disease was able to induce hyperthyroidism in experimental animals. LATS were later found to be thyroid-stimulating hormone receptor (TSHR)-stimulating antibodies.
TSHR is a membrane receptor that belongs to a superfamily that contains hormone receptors for LH, FSH, and HCG. The extracellular domain (ectodomain, subunit A) of TSHR has several leucine-rich repeats (LRDs) that create a binding site for thyroid-stimulating hormones (TSH). Anti-TSHR stimulating and blocking autoantibodies bind to the same domain though with different Fab fragment orientations, the latter accounting for different downstream cellular events. TSHR stimulating antibodies (Abs) may have a higher affinity for the TSH binding site on TSHR and even displace TSH, thereby inducing unremitting thyroid hormone production and thyrocyte proliferation without being controlled by the physiological feedback mechanism. The latter antibodies typically belong to the IgG1 subclass.1–10 A well-characterized human monoclonal antibody (m22) from patients with GD was found to have TSHR-stimulating activity with high affinity (circa 6.7 × 10-11 mol/L).8 The tests based on this antibody offer high clinical sensitivity and specificity.
Diagnosis of Grave’s disease is based on clinical grounds and on blood levels of free T3, free T4, and thyroid-stimulating immunoglobulins. This, however, can be aided by thyroid-stimulating immunoglobulins testing in certain circumstances, such as: i) when Grave’s orbitopathy (GO) or dermopathy is absent, but other results point to Grave’s disease; ii) in pregnant women with Grave’s disease; and iii) in patients on antithyroid drugs (ATDs) to determine if the medication can be discontinued.
It is important to note that Grave’s disease and Hashimoto thyroiditis are two extremes of an autoimmune spectrum: over time, patients may move from one extreme to the other or, at times, manifest a fluctuating course. There are TSHR-blocking Abs and anti-TSHR Abs that only bind to other parts of TSHR with different affinities; these may be of additional subclasses, such as IgG2 and IgG3. It is not uncommon to see patients with either Grave’s disease or Hashimoto thyroiditis that have both stimulating and blocking antibodies at the same time.
It appears that the titer and the avidity of the Ab population determine the outcome in some circumstances; however, it is pivotal to note that the final diagnosis and management decisions should not be solely based on TSI test results. What is more important than a qualitative TSI test result is the measurement of TSI concentrations in patients over a period of time; for instance, TSI is found in 7% of Hashimoto thyroiditis patients without Grave’s orbitopathy, but in 68% of Hashimoto thyroiditis patients with Grave’s orbitopathy.1–10
Additionally, by using blocking Ab bioassay, the prevalence of TSHR-blocking Abs among Grave’s disease and Hashimoto thyroiditis patients is established around 4% and 9%, respectively.11 Approximately 96% of Grave’s disease patients do not have TSHR-blocking Abs, thus obviating the need for distinguishing between the stimulatory and blocking antibodies in these patients. This avoids the costly and labor-intensive nature of bioassay without making a significant difference in patient management.
Methodology
The IMMULITE® 2000/2000 XPi TSI assay (Siemens) is FDA-approved for in vitro diagnostic purposes.
The test principle is based on automated, random-access, two-cycle chemiluminescent bridging immunoassay. The solid phase (polystyrene beads) are coated, through a monoclonal Ab, with a chimeric human TSHR (Mc4). In Mc4, the TSH binding site is intact, but its membrane-proximal part has been replaced by rat luteinizing hormone-choriogonadotropin receptor to not only stabilize the molecule so it can maintain its native form but also to preclude “other” TSHR-binding (but non-stimulating) Abs of binding to this chimeric molecule. Mc4 is also used by a commercial bioassay. During the second step, the capture TSHR is added, but the latter is conjugated to alkaline phosphatase (ALP), which will generate the chemiluminescent signal upon the addition of the substrate. The generated photons are read as relative light units, or counts per second, for calculations (Figure).
The kit uses two levels (low, high) of “adjustors” in replicates of four (logistical model). These are m22 Abs, as described above, and are used as calibrators based on which the final TSI concentration is calculated in international units per liter (IU/L).
The calibration is based upon a lot-specific master curve generated by the manufacturer. By using such adjustors and the test design, the stimulatory antibodies are typically measured. Furthermore, it has been demonstrated that stimulatory antibodies, such as m22, are able to bind to the concave region of TSHR LRDs where TSH also binds, whereas the blocking antibodies, such as K1-70, bind to the receptor more N-terminally and 155˚ away from where stimulatory antibodies bind.12 This key difference helps this kit to significantly avoid bridging the receptors by blocking antibodies due to steric hindrance, the latter of which is further accentuated by the upside-down nature of the signal receptor and presence of the conjugated ALP.
According to the manufacturer, this assay is traceable to WHO 2nd International Standard for thyroid-stimulating antibody, National Institute for Biological Standards and Control (NIBSC), code: 08/204.
The kit also has three controls (negative, low, and high-positive) using m22 Ab. The reportable range is 0.10-40 IU/L, but the instrument can do further dilutions to calculate the final concentration. The manufacturer recommends 0.55 IU/L as the positivity cut-off based on which the clinical sensitivity and specificity are 98.6% and 98.5%, respectively. This is congruent with the most recent American Thyroid Association (ATA) guideline that states:
In the setting of overt thyrotoxicosis, newer TRAb binding and bioassays have a sensitivity of 96–97% and a specificity of 99% for GD. 7
It is noteworthy to mention that ATA did not specifically mention this test, and by “TRAb”, they referred to the two commercial ELISAs (TBI). TSI Immulite received FDA approval after the guideline was finalized. It was also previously demonstrated that TSI Immulite test had higher sensitivity than bioassay in patients with GD on ATDs.9 Furthermore, the manufacturer claims that IMMULITE® 2000/2000 XPi TSI test results are not affected in sera spiked by high concentrations of FSH, LH, TSH, HCG, anti-thyroglobulin, and anti-thyroid peroxidase.
Last but not least, a bioassay only measures cyclic adenosine monophosphate (cAMP); however, other TSHR activation pathways that do not result in cAMP production, such as phospholipase C cascade, are not assessed using current bioassays. This is even of more importance when, in Grave’s orbitopathy, IGF-1 receptor forms a complex with TSHR to trigger orbital fibroblasts for glycosaminoglycan production and lipogenesis.6
Advantages
The IMMULITE® 2000/2000 XPi TSI assay analytically and clinically maintains high levels of quality and performance characteristics while significantly lowering the reagent cost and technologist’s time. In comparison, a bioassay requires cell culture, multiple reagents, ancillary tests, and manual calculations, all of which predispose this test to analytical and post-analytical errors, QC failures, lack of amenability to automation, and extreme labor intensiveness. These factors adversely affect the cross-training of new technologists.
This test is performed at Cleveland Clinic on a random-access platform, Monday through Friday. Tests can be added at any time during working hours without the need for batch-testing, obviating the prolonged bioassay turnaround time, as the former only takes 65 minutes to complete. This walk-away system immensely spares the laboratory technician’s time.
While Cleveland Clinic currently utilizes the IMMULITE®2000 system, another random-access, commercially-available platform is Elecsys® Anti-TSHR (Roche). However, in addition to the published reports9, 10 that show relatively poor performance, the test principle ultimately makes this option a sub-optimal choice: porcine TSH (instead of the Mc4 construct), murine monoclonal antibodies (that make heterophile Ab interference likely), the utilization of biotin (for conjugation of the latter Abs that make the test prone to falsely-elevated results due to dietary biotin), combined together argue against its usefulness for TSI testing.
Upon discussion with several other laboratories in the United States that have adopted the IMMULITE® 2000/2000 XPi TSI assay, each expressed high satisfaction.
Validation Summary
To verify the manufacturer’s claims, the TSI Immulite MMULITE® 2000/2000 XPi TSI test was validated in-house within the Immunopathology Laboratory on Cleveland Clinic’s main campus.
The linearity and accuracy study used a neat sample and multiple dilutions up to 1:250 in triplicate. The recovery rate, slope, intercept, and observed error were all within acceptable and established limits. For the precision study, the inter- and intra-run simple and complex precision was performed using low, medium, and high-level samples. The %CV values all were well within acceptable limits, confirming high precision, which is ideal for monitoring patients over time.
The method comparison study was performed in two parts:
- One was performed using known positive sera (70% female, age: 24–78 years) received from ARUP Laboratories. ARUP utilizes an in-house-developed bioassay. Our laboratories received samples within 128-480% (low to high positive); their test uses 123% as the positivity cut-off.
- The second part was performed using sera received from the Ohio State University Laboratories, as they also utilize the IMMULITE® 2000/2000 XPi TSI assay.
- Both panels showed 100% categorical agreement, confirming comparability.
According to our alternate proficiency testing results, our in-house bioassay had 100% concordance with the ARUP in-house-developed bioassay.
A carryover study used three replicates in low, high, low order, and three replicates of a high positive sample with 1:10 dilution. The results confirmed no significant instrument carryover.
The reference range study used apparently healthy subjects, as none tested positive for TSI (their TSI results were all <0.10 IU/L). This verified the manufacturer positivity cut-off of 0.55 IU/L.
References
1. Frank CU, Braeth S, Dietrich JW, Wanjura D, Loos U. Bridge Technology with TSH Receptor Chimera for Sensitive Direct Detection of TSH Receptor Antibodies Causing Graves’ Disease: Analytical and Clinical Evaluation. Horm Metab Res. 2015 Nov;47(12):880-8. doi: 10.1055/s-0035-1554662. Epub 2015 Jun 16.
2. McLachlan SM, Rapoport B. Thyrotropin-blocking autoantibodies and thyroid-stimulating autoantibodies: potential mechanisms involved in the pendulum swinging from hypothyroidism to hyperthyroidism or vice versa.
3. Nguyen CT, Sasso EB2, Barton L, Mestman JH. Graves’ hyperthyroidism in pregnancy: a clinical review. Clin Diabetes Endocrinol. 2018 Mar 1;4:4. doi: 10.1186/ s40842-018-0054-7. eCollection 2018.
4. Bitcon V, Donnelly J, Kiaei D. Sensitivity of assays for TSH-receptor antibodies. J Endocrinol Invest. 2016 Oct;39(10):1195-6. doi: 10.1007/s40618-016-0520-y. Epub 2016 Aug 16.
5. Tozzoli R, D’Aurizio F, Villalta D, Giovanella L. Evaluation of the first fully automated immunoassay method for the measurement of stimulating TSH receptor autoantibodies in Graves’ disease. Clin Chem Lab Med. 2017 Jan 1;55(1):58-64. doi: 10.1515/cclm-2016-0197.
6. Smith TJ, Hegedüs L. Graves’ Disease. N Engl J Med. 2016 Oct 20;375(16):1552-1565.
7. Ross DS, Burch HB, Cooper DS, Greenlee MC, Laurberg P, Maia AL, Rivkees SA, Samuels M, Sosa JA, Stan MN, Walter MA. 2016 American Thyroid Association Guidelines for Diagnosis and Management of Hyperthyroidism and Other Causes of Thyrotoxicosis. Thyroid. 2016 Oct;26(10):1343-1421.
8. Sanders J, Evans M, Premawardhana LD, Depraetere H, Jeffreys J, Richards T, Furmaniak J, Rees Smith B. Human monoclonal thyroid-stimulating autoantibody. Lancet. 2003 Jul 12;362(9378):126-8.
9. David J. Kemble, Tara Jackson, Mike Morrison, Mark A. Cervinski, Robert D. Nerenz. Analytical and Clinical Validation of Two Commercially Available Immunoassays Used in the Detection of TSHR Antibodies. DOI: 10.1373/jalm.2017.024067 Published October 2017.
10. Y Li, J Kim, T Diana, R Klasen, P D Olivo, and G J Kahaly. A novel bioassay for anti-thyrotrophin receptor autoantibodies detects both thyroid-blocking and stimulating activity. Clin Exp Immunol. 2013 Sep; 173(3): 390–397.
11. Diana T, Krause J, Olivo PD, König J, Kanitz M, Decallonne B, Kahaly GJ. Prevalence and clinical relevance of thyroid-stimulating hormone receptor-blocking antibodies in autoimmune thyroid disease. Clin Exp Immunol. 2017 Sep;189(3):304-309. doi: 10.1111/cei.12980.
12. Sanders P1, Young S, Sanders J, Kabelis K, Baker S, Sullivan A, Evans M, Clark J, Wilmot J, Hu X, Roberts E, Powell M, Núñez Miguel R, Furmaniak J, Rees Smith B. Crystal structure of the TSH receptor (TSHR) bound to a blocking-type TSHR autoantibody. J Mol Endocrinol. 2011 Feb 15;46(2):81-99. doi: 10.1530/JME-10-0127. Print 2011 Apr.