Special Communication
New Chemistry Standard Transport Tube
New Chemistry Standard Transport Tube Implemented for All Cleveland Clinic Locations
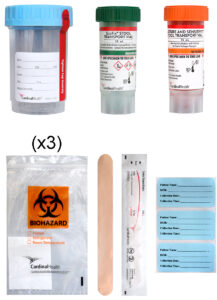
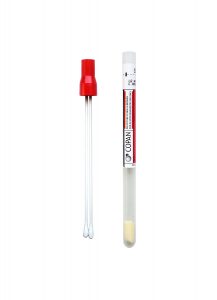
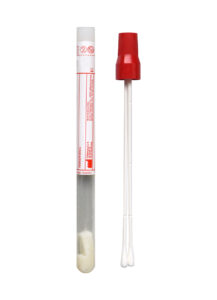
A new standard transport tube will be implemented for all Cleveland Clinic locations and clients on March 9, 2026. This new tube will replace the existing transport tube (Sarstedt product number 62.612) for most specimens that need to be aliquoted. The existing transport tube will no longer be accepted after March 31. Please refer to the test directory or procedure catalog for test-specific requirements.