Category: Special Communication
February 2021: Inhibin B – Changes to Reference Ranges
Special Communication
Inhibin B – Changes to Reference Ranges
Effective February 16, 2021, methodology and reference intervals for Inhibin B (INHIBB) will change.
Test results obtained with the new ANSH Ultra-Sensitive Inhibin B ELISA method and the previous Beckman Coulter Inhibin G Gen II ELISA method cannot be used interchangeably.
Beginning February 16 through May 16, 2021, specimens will be analyzed with both methods to establish a new baseline for individual patient results. Results from the previous method and reference intervals will be posted in a comment on the patient’s chart.
This change increases the detection of inhibin B in all clinically relevant cases and expands the testing range to improve sensitivity.
Additional details are available in the February 2021 Technical Update.
Changes to Reference Ranges
Inhibin B (INHIBB)
Female
1 day – 12 years:
1 – 182 pg/mL
13 – 41 years (regular cycle, follicular phase):
8 – 223 pg/mL
42 – 51 years (regular cycle, follicular phase):
1 – 107 pg/mL
51 – 76 years (postmenopausal):
1 – 11 pg/mL
Male
< 15 days:
68 – 373 pg/mL
15 days – 6 months:
42 – 516 pg/mL
7 months – 7 years:
24 – 300 pg/mL
8 – 30 years:
47 – 383 pg/mL
31 – 72 years:
10 – 357 pg/mL
Methodology
ANSH ultrasensitive Inhibin B ELISA method
formerly Beckman Coulter Inhibin B Gen II ELISA method
November 2020: Temporarily Unavailable – Stool Gastrointestinal Panel by PCR
Special Communication
Temporarily Unavailable: Stool Gastrointestinal Panel by PCR
Beginning November 13, 2020, Stool Gastrointestinal Panel by PCR (STGIPR) testing is temporarily unavailable to order from CCL.
Cleveland Clinic Laboratories is experiencing severe supply constraints for Stool Gastrointestinal Panel by PCR (STGIPR) testing. These shortages impact laboratories nationwide, resulting in the inability to identify a reference laboratory to forward this testing.
Alternative Testing Options
The tests listed below cover the range of pathogens detected in the Stool Gastrointestinal Panel by PCR (STGIPR) test. Depending on clinical circumstances and judgment, various combinations of these tests may serve as an appropriate substitution for STGIPR:
Test Code
Components
Performing Lab
STLPCR
Salmonella spp., Shigella spp., Campylobacter jejuni/coli, and Shiga-toxin (stx1 and stx2) genes
CCL
OVAPSC
Giardia lamblia and Cryptosporidium species
CCL
CRYSPO
Cryptosporidium, Cyclospora, and Cystoisospora sp.
CCL
OVAP
Cryptosporidium and Giardia
CCL
CDPCR
C. difficile toxin B gene
CCL
EROTA
Rotavirus antigen
CCL
VIBCUL
Vibrio
CCL
YERCUL
Yersinia
CCL
NORPCR
Norovirus 1 & 2
ARUP
SADNO
Adenovirus antigen
Focus
COVID-19: Supply Constraints Affecting Enteric Bacterial Panel by PCR Testing
Special Communication
Supply Constraints Affecting Enteric Bacterial Panel by PCR Testing
Due to supply constraints related to COVID-19, supplies for Enteric Bacterial Panel by PCR (STLPCR) testing are in extremely limited supply.
While supplies are constrained, culture and antigen testing will be used in place of PCR testing to interrogate specimens for the same pathogens as STLPCR: Campylobacter, Salmonella, Shigella, and Shiga toxin-producing E. coli (STEC/EHEC).
Cleveland Clinic Laboratories will temporarily cancel and credit any STLPCR test requests, then substitute clinically-equivalent culture and antigen testing.
The associated CPT codes for these substitutions are:
• 87449 (x 2)
• 87045

COVID-19: Unacceptable Specimen Transport Media for COVID-19 Testing
Special Communication
Unacceptable Specimen Transport Media for COVID-19 Testing
Due to incompatibilities with our COVID-19 testing laboratory equipment, Cleveland Clinic Laboratories has determined that specimens in the following media are not acceptable for COVID-19 testing performed by CCL:


The following specimen types are accepted by CCL for COVID-19 testing:




Universal Transport Media (UTM), 3 mL
Viral Transport Medium (VTM), 3 mL
Saline Transport Media, 3 mL
Sterile Container*, 3 mL
Specimen Type:
• Nasopharyngeal (NP) swab – preferred
• Nasal (anterior nares) swab
• Oropharyngeal (OP) swab
Specimen Type:
• Nasopharyngeal (NP) swab – preferred
• Nasal (anterior nares) swab
• Oropharyngeal (OP) swab
Specimen Type:
• Nasopharyngeal (NP) swab – preferred
• Nasal (anterior nares) swab
• Oropharyngeal (OP) swab
Specimen Type:
• Sputum
• Bronchoalveolar lavage (BAL)
*lower respiratory specimens only
Want More Information about COVID-19 Testing?
Review our Coronavirus 2019 Testing Overview for additional details, including collection instructions, required patient demographics, and more.
Need Collection & Transport Media?
If you are having difficulties obtaining appropriate COVID-19 specimen collection and transport supplies, please contact your CCL Account Manager.
Have a Question?
Please contact Client Services at 800.628.6818.
COVID-19: Supply Shortages Impacting Gonorrhea, Chlamydia, and Trichomoniasis Testing
Special Communication
Supply Shortages Impacting Gonorrhea, Chlamydia, and Trichomoniasis Testing
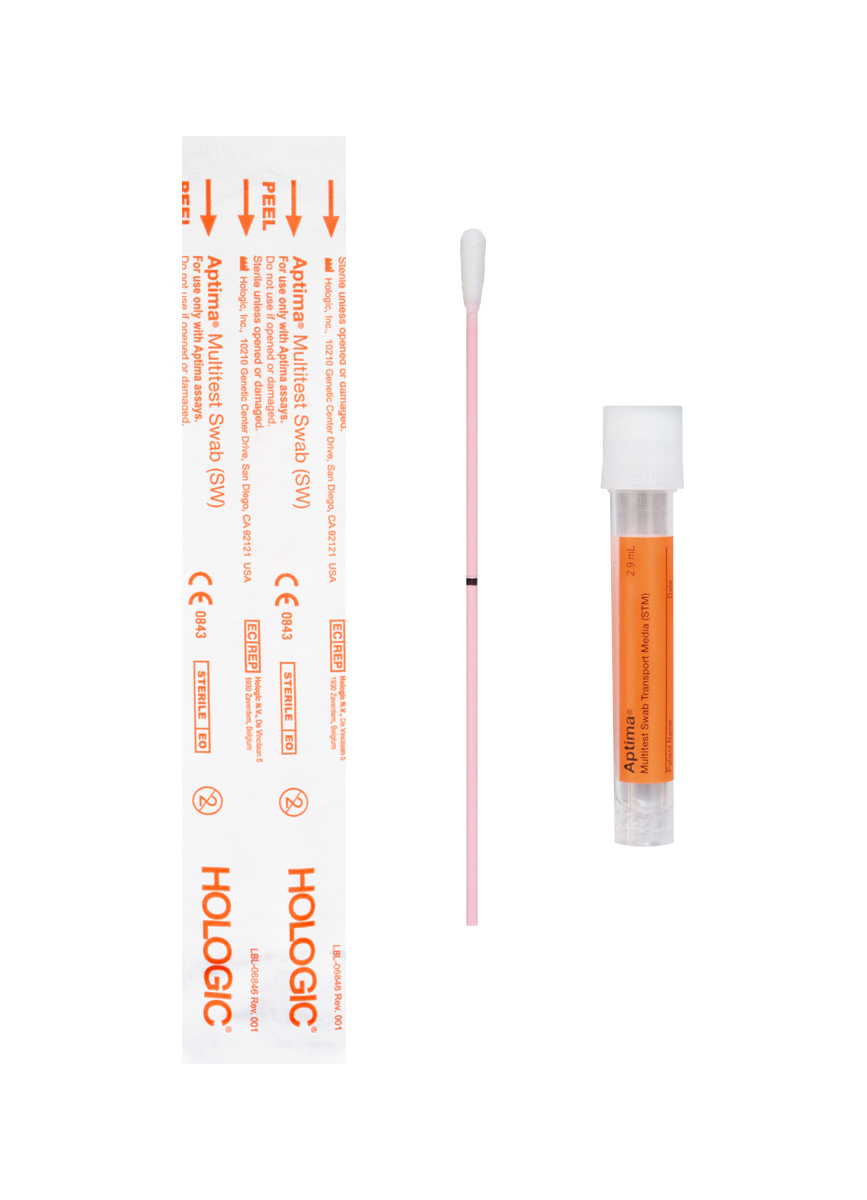
As a result of nationwide supply shortages caused by COVID-19 demands, Cleveland Clinic Laboratories may periodically be unable to provide Aptima® Urine Specimen Collection Transport Tubes for gonorrhea, chlamydia, and trichomoniasis testing.
Aptima tubes will continue to remain an acceptable specimen collection container for testing.
Because of the severe shortage of urine tubes, providers should reserve urine testing for men with persistent urethritis. This practice aligns with guidance from the Centers for Disease Control and Prevention (CDC).

What Tests Are Affected
GC/Chlamydia Amplification, Urine (UGCCT)
Ordering Options for Gonorrhea/Chlamydia Testing
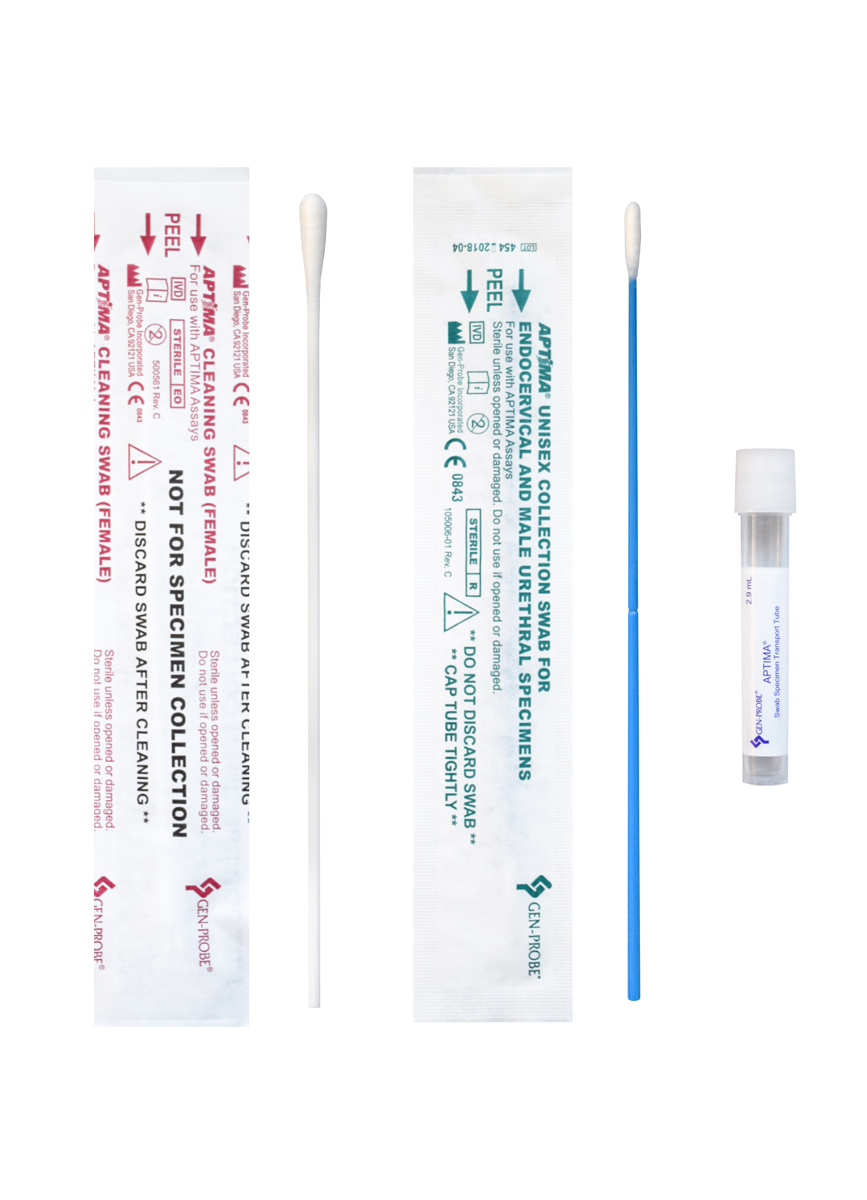
- GC/Chlamydia Amplification, Urine (UGCCT) using self-supplied Aptima Urine Specimen Collection Transport Tubes
- GC/Chlamydia Amplification, Genital, Rectal and Oral Specimens (GCCT) using an Aptima Swab Kit
Trichomonas vaginalis Amplification (TRVAMP)
Ordering Options for Trichomoniasis Testing
- Trichomonas vaginalis Amplification (TRVAMP) using an Aptima Swab Kit
Specimen Collection for GCCT & TRVAMP
Samples can continue to be collected and submitted with either Aptima kit shown below.
Although CCL cannot provide this item, Aptima tubes will continue to remain an acceptable specimen collection container for testing.
Unacceptable Specimen Containers
Because of the limited stability of unpreserved urine for gonorrhea, chlamydia, and trichomoniasis testing, Cleveland Clinic Laboratories is currently unable to accept urine in a sterile container (i.e., specimen cup) for UGCCT and TRVAMP orders.

Other Considerations
Cleveland Clinic Laboratories will notify clients when Aptima urine tubes are back in stock and available to order. Conservative ordering practices may be necessary until supplies stabilize.
For help in prioritizing which patients to test for these pathogens, please refer to the recently-issued considerations from the CDC.
August 2020: Update – Cytogenetic Karyotyping & Chromosomal Microarray Testing
Special Communication
Update: Cytogenetic Karyotyping & Chromosomal Microarray Testing
Cleveland Clinic Laboratories (CCL) remains committed to supporting patients and providers during the COVID-19 pandemic.
To meet the current demands for COVID-19 PCR testing, Cleveland Clinic Laboratories is temporarily reassigning some of our molecular pathology technologists to this effort.
As a result, cytogenetic karyotyping and chromosomal microarray testing will temporarily be sent out to a partnered reference laboratory. This will allow COVID-19 testing to continue uninterrupted while we increase our testing capacity.
What Tests Are Affected
Starting Monday, August 24, 2020, Cleveland Clinic Laboratories will begin temporarily sending these tests to Quest Diagnostics.
Test Name
CCL Test Code
Send-out Test Code (Quest)
Expected TAT
Chromosome Analysis, Bone Marrow w/ Reflex SNP Array
9 Days
13 Days
Chromosome Analysis, Solid Tumor (Lymph Nodes)
22 Days
11 Days
Test Name
CCL Test Code
Send-out Test Code (Quest)
Expected TAT
Timing
Starting Monday, August 24, 2020, Cleveland Clinic Laboratories will begin temporarily sending these tests to Quest Diagnostics.
CCL estimates that this process will continue for several months. We will return these tests in-house as soon as possible and communicate any changes once they are available.
Turnaround Time
In some instances, we anticipate that turnaround times (TAT) may be impacted. CCL will do everything possible to minimize delays.
Ordering & Results
There are no changes to the ordering process.
Test results will continue to be available via the Atlas portal or faxed results. Please contact your CCL Sales representative as necessary for further support in retrieving these results.
Billing
At this time, we do not anticipate any changes to the billing process.
Pricing will reflect costs associated with sending out these tests. A Cleveland Clinic Laboratories representative will communicate any price changes directly to clients.
August 2020: Anti-cardiolipin IgM, IgA, and IgG Reporting
Special Communication
Anti-cardiolipin IgM, IgA, and IgG Reporting
Effective September 3, 2020, Cleveland Clinic Laboratories will report Anti-cardiolipin IgM, IgA, and IgG semi-quantitative enzyme immunoassays with updated reference ranges.
This change is in accordance with the manufacturer’s instructions (Inova Diagnostics), as approved by the FDA.
Anti-cardiolipin antibody testing aids in diagnosing antiphospholipid syndrome or assessing the risk of thrombosis in individuals with systemic lupus erythematosus, among others.
In addition to reference range changes, the manufacturer has recently replaced the serum calibrators and controls prepared from patients’ sera used in the anti-cardiolipin IgM kit with a monoclonal recombinant antibody suspended in negative human serum. This change will improve performance, stability, and lot-to-lot consistency.
Changes to Reference Ranges
Cardiolipin IgM Antibodies (CARDIM)
Negative:
< 12.5 MPL
Indeterminate:
12.5 – 20 MPL
Positive:
> 20 MPL (98.2th percentile)
Cardiolipin IgA Antibodies (CARDIA)
Negative:
< 12.0 APL
Indeterminate:
12 – 20 APL
Positive:
> 20 APL (100th percentile)
Cardiolipin IgG Antibodies (CARDIG)
Negative:
< 15.0 GPL
Indeterminate:
15 – 20 GPL
Positive:
> 20 GPL (98.6th percentile)
Panels Affected
Cardiolipin Antibodies (CARDIO)
Cardiolipin Antibodies (CARDIO) is comprised of the three tests listed above.
Hypercoagulation Diagnostic Interpretive Panel (HYPER)
This panel will be impacted by the changes to the above three tests.
Lupus Anticoagulant Diagnostic Interpretive Panel (LUPUSP)
This panel will be impacted by the changes to the above three tests.
Antibody Testing for COVID-19: Overview & FAQs
CAP Statement on the Current Role of Serologic Testing for SARS-CoV-2
May 13, 2020 | From the College of American Pathologists
Understanding COVID-19 & Antibody Testing
May 7, 2020 | From Cleveland Clinic’s Health Essentials:
We’ve accepted the harsh realities of COVID-19 and we’ve adjusted our lives accordingly. And yet, we still have so many unanswered questions like why has COVID-19 taken such a detrimental toll, and where are we in the process of finding a cure?
To get a better idea of where science is headed, Cleveland Clinic CEO and President Tom Mihaljevic, MD interviews Serpil Erzurum, MD, Chair of the Lerner Research Institute.
COVID-19 Serologic Testing: FAQS and Caveats
April 29, 2020 | From the Cleveland Clinic Journal of Medicine’s COVID-19 Curbside Consults:
There has been an immense amount of tangential discussion regarding the potential usefulness of serologic testing for COVID-19 recently. Although serologic testing has never been routinely used for diagnosing infections with “respiratory viruses”, such as influenzae, parainfluenzae, respiratory syncytial viruses, adenoviruses, or metapneumovirus, questioning its usefulness during the current outbreak has relevance.
That being said, during global epidemics of SARS, MERS, and H1N1 influenza, serology was never used in routine diagnostics. However, given the pandemic status of COVID-19 and the shortage of nucleic acid detection kits and/or swabs in certain areas, it raises the prospect of resorting to serology as an alternative to directly testing for the presence of the virus. The Infectious Diseases Society of America (IDAS) has recently issued a clear statement on COVID-19 serology.
COVID-19: Laboratory Resources
Respiratory Virus Testing
Updated October 2025
Respiratory Virus Testing
Cleveland Clinic Laboratories offers several respiratory testing services to healthcare organizations & providers.
SARS-CoV-2 (COVID-19), influenza A/B, and respiratory syncytial virus (RSV) testing is available for the qualitative detection of these viruses in respiratory specimens from individuals with appropriate signs, symptoms, and risk factors.
All three viruses are expected to be in circulation during the 2025-2026 respiratory virus season.
Respiratory Virus Testing Overview
Test Name
COVID, Influenza A/B, & RSV NAAT, Routine (CVFLRS)
Turnaround Time
24 hours from the time of receipt by the laboratory*
Testing is performed 24 hours a day, 7 days a week
Specimen Requirements – Upper Respiratory
Type:
(1) Nasopharyngeal (NP) Swab – preferred
Volume:
3 mL
Specimen Container:
Universal Transport Media (UTM)
Accepted Alternatives
Type:
• (1) Nasopharyngeal (NP) swab
• (1) Nasal (anterior nares) swab
Volume:
Swab
Transport Media:
• Universal Transport Media (UTM)
• eSwab
• Saline (sterile)
• Viral Transport Media (VTM), including M4RT, M5, or M6
Specimen Requirements – Lower Respiratory
Type:
• Aspirate, tracheal
• Bronchoalveolar Lavage (BAL)
• Sputum
Volume:
1 mL
Transport Container:
Sterile container

Transport Temperature
Refrigerated; transport to the laboratories on cold packs or wet ice.
Stability
Ambient:
Unacceptable
Refrigerated (2-8ºC):
96 hours
Frozen:
Up to 30 days
Rejection Criteria
Expanded Respiratory Pathogen Panel Test Overview
Test Name
Expanded Respiratory Pathogen Panel by PCR (with COVID), Routine (RPPCR)
Turnaround Time
2 – 3 days
Specimen Requirements – Upper Respiratory
Type:
(1) Nasopharyngeal (NP) Swab – preferred
Volume:
3 mL
Specimen Container:
Universal Transport Media (UTM)

Accepted Alternatives
Type:
• (1) Nasopharyngeal (NP) swab
• (1) Nasal (anterior nares) swab
Volume:
Swab
Transport Media:
• Universal Transport Media (UTM)
• eSwab
• Saline (sterile)
• Viral Transport Media (VTM), including M4RT, M5, or M6
Specimen Requirements – Lower Respiratory
Type:
• Aspirate, tracheal
• Bronchoalveolar Lavage (BAL)
• Sputum
Volume:
1 mL
Transport Container:
Sterile container

Transport Temperature
Refrigerated; transport to the laboratories on cold packs or wet ice.
Stability, Upper Resp.
Ambient:
Unacceptable
Refrigerated (2-8ºC):
3 days
Frozen:
Up to 30 days
Stability, Lower Resp.
Ambient:
Unacceptable
Refrigerated (2-8ºC):
7 days
Frozen:
Up to 30 days
Rejection Criteria
Ready to Place an Order?
Download and complete a Respiratory Virus Testing Requisition (required).
Need to Set Up an Account?
Request support from a regional Account Manager or contact Laboratory Customer Service for general inquiries.
Specimen Collection Instructions
You will need:
• Personal protective equipment (PPE)
• (1) tube of transport medium
– Universal Transport Media (UTM)
– Viral Transport Media (VTM)
– Saline solution
• (1) swab
• Patient identification labels
Before you begin:
• Confirm the patient’s name and date of birth.
• Wash your hands, then put on personal protective equipment, including gloves.
Nasopharyngeal (NP) Swabs
Step 1:
Carefully remove the swab from the packaging without touching the tip.
Step 2a – for nasopharyngeal (NP) swabs:
– Gently insert the NP swab along the nasal septum just above the floor of the passage to the nasopharynx until resistance is met.
– Rotate the swab gently against the nasopharyngeal mucosa for 10 – 15 seconds, then remove.
Step 3:
Remove the cap from the tube, then insert the swab all the way to the bottom of the vial.
Step 4:
While holding the tube away from your face, bend the swab in the vial at a 180-degree angle to break at the marked breakpoint.
Step 5:
Close the tube tightly. If possible, wrap the cap with Parafilm.
Step 6:
Label specimen with the printed label or write the patient’s first & last name, date of birth, and the date, time, and site of collection (e.g., NP) on the tube.
Step 7:
Double-bag the specimen, place on wet ice, then refrigerate.
Step 8:
Complete a Respiratory Virus Test Requisition.
Step 9:
Place the completed requisition in the pocket of the specimen bag.
Step 10:
For the fastest turnaround time, deliver the specimen directly to the Main Campus LL Building Specimen Receiving Area:
Cleveland Clinic Laboratories
10300 Carnegie Avenue
Cleveland, OH 44106
Nasal (Anterior Nares) Swabs
Specimen Collection:
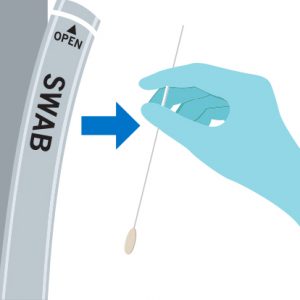
Step 1:Carefully remove the swab from the packaging without touching the tip.
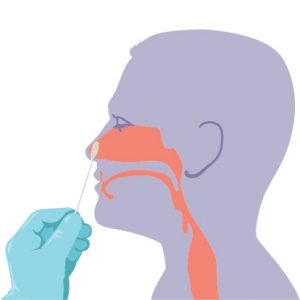
Step 2:
Position the patient’s head slightly back, then insert the swab into their LEFT nostril (less than one inch) and gently push until a slight resistance is met.
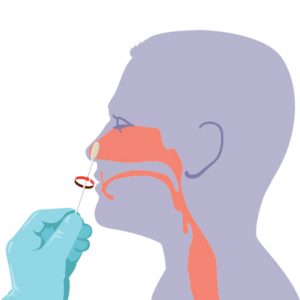
Step 3:
Rotate the swab against the inside of the patient’s nostril four times for 10-15 seconds.
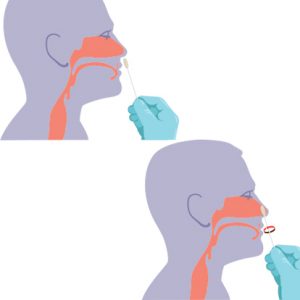
Step 4:
Remove the swab, then repeat Steps 2 & 3 in the RIGHT nostril with the same swab.
Remove the swab from the patient’s nose when finished.
Preparing for Transport:
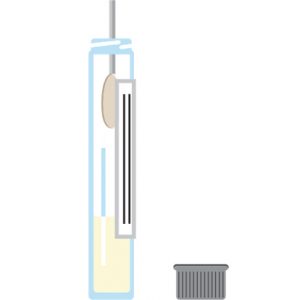
Step 5:
While holding the swab, open the tube and set the cap down with the threads facing up.
Insert the swab into the tube until the breakpoint is level with the tube opening.
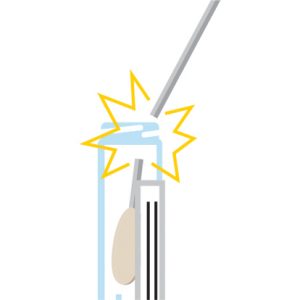
Step 6:
While holding the tube away from your face, bend the swab until it snaps at the break point.
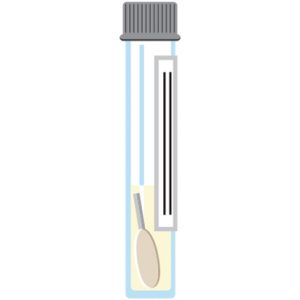
Step 7:
After discarding the remaining swab shaft into an approved disposal container, screw the cap back onto the tube.
Apply the test label or write the patient’s information onto the tube.

Step 8:
Place the specimen into a biohazard bag, then insert any lab paperwork into the outside pouch.
Resources
COVID-19 Resources
Influenza Resources
RSV Resources
If you have any questions, please contact your CCL Account Manager or Laboratory Customer Service for assistance.