Special Communication
Changes to NCYTO, URCAT2, MYOSPL, RUFIN, LIVFIB, & AJPWO Testing
Changes to Specimen Requirements
Anaplasma phagocytophilum (HGA) Antibodies, IgG and IgM (ANIGM)
Specimen Requirements
Specimen Type:
Serum
Volume:
0.5 mL
Minimum Volume:
0.2 mL
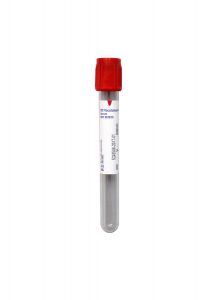
Collection Container:
Gold Serum Separation Tube (SST)
Transport Temperature:
Refrigerated
Babesia Microti IgG & IgM Abs (BMICGM)
Specimen Requirements
Specimen Type:
Serum
Volume:
1 mL
Minimum Volume:
0.2 mL
Collection Container:
Gold Serum Separation Tube (SST)
Transport Temperature:
Refrigerated
Additional Information:
Remove serum from cells immediately or within 2 hours of collection. Parallel testing is preferred.
Convalescent specimens must be received within 30 days from receipt of the acute specimens. Please mark specimens as “acute” or “convalescent”.
Copper: Free, Serum, or Plasma (FRCOP)
Specimen Requirements
Specimen Type:
Serum
Volume:
3 mL
Minimum Volume:
1.2 mL
Collection Container:
Royal Blue (Red Stripe), Plain – Trace Metal-Free
Transport Temperature:
Refrigerated
Alternative Specimen:
Plasma
Alternative Container:
Royal Blue (Purple Stripe), with K2EDTA – Trace Metal-Free
Additional Information:
Do not use separator tubes. Remove from cells immediately and aliquot into a trace metal-free transport tube (ARUP #43116) or acid-washed transfer vial (ARUP #54350).
Changes to Reference Ranges
Catecholamines Fractionated by LC-MS/MS, Urine Free (URCAT2)
There will be no other reference range changes for URCAT2.
Norepinephrine, Urine 24h
0 – 3 Years:
Not Established
4 – 10 Years:
7 – 65 µg/d
11 – 17 Years:
12 – 96 µg/d
18 – 99 Years:
14 – 120 µg/d
Dopamine, Urine 24h
0 – 3 Years:
Not Established
4 – 10 Years:
80 – 440 µg/d
11 – 17 Years:
100 – 496 µg/d
18 – 99 Years:
71 – 485 µg/d
Cystine, Urine Quant (UCYSTD)
0 – 2 Months:
≤ 870 µmol/g crt
3 – 11 Months:
≤ 300 µmol/g crt
1 – 2 Years:
≤ 150 µmol/g crt
3 – 5 Years:
≤ 125 µmol/g crt
6 – 11 Years:
≤ 100 µmol/g crt
12 Years & Older:
≤ 150 µmol/g crt
Purine and Pyrimidine Panel (UPURPY)
There will be no other reference range changes for UPURPY.
S-Sulfocysteine
0 – 3 Years:
≤ 11 mmol/mol Cr
4 – 6 Years:
≤ 5 mmol/mol Cr
7 – 12 Years:
≤ 5 mmol/mol Cr
13 – 18 Years:
≤ 5 mmol/mol Cr
18 Years & Older:
≤ 5 mmol/mol Cr
Any additional information will be included in a future Technical Update. If you have any questions about these changes, please contact Client Services for assistance.